 
Industrial processes generate a variety of molecules that may pollute waters due to negative impacts for ecosystems and humans (toxicity, carcinogenic, and mutagenic properties). 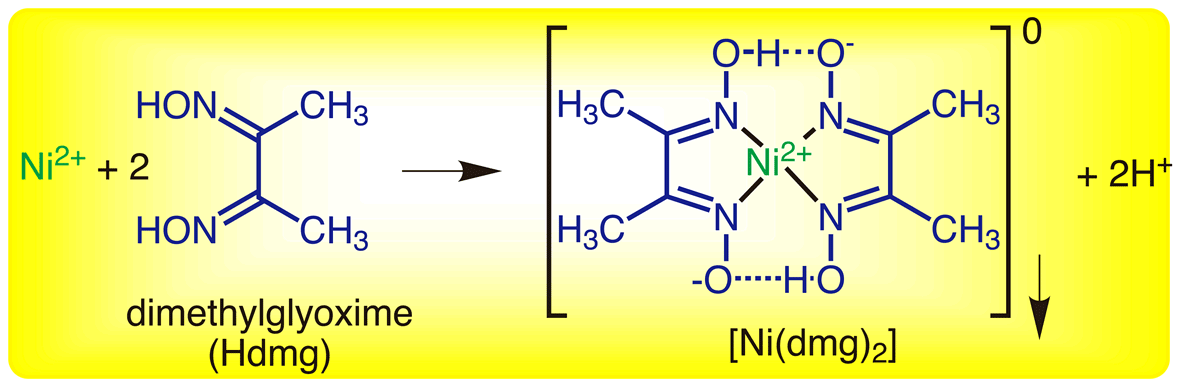 
The amounts of several pollutants present in the surface and underground waters have increased in the last years. The degradation process obeys the first-order kinetic. The Ni-DMG particles out of zeolite framework did not show significant degradation efficiency. The optimal operation parameters were found as follows:, temperature of 60☌, 0.6 g L −1 of the catalyst, and 40 ppm of the dye concentration. UV-Vis spectrophotometric measurements were performed for determination of the decolorization and mineralization extents. The effect of key operating parameters such as catalyst dosage, temperature, the initial concentration of the dye, and pH of the samples was studied on the degradation extent of the dye. The prepared composite was used as a catalyst in the photodegradation process of an aqueous solution methyl green (MG) dye under UV irradiation. FT-IR, XRD, SEM, TG, and DTG methods were used for characterization of the raw and modified samples. Ni-DMG/ZSM-5 zeolite was prepared by ion exchange and complexation procedures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
